In comatose adults who present to the emergency department is administration of thiamine necessary before glucose for prevention of Wernicke Korsakoff Syndrome Clinical Scenario A 45 year old homeless man presents to the emergency department after he was found lying on a park bench. Glasgow Coma Scale is 5, so he is intubated. Because of thiamine’s role in glucose metabolism, IV glucose or PN infusion can precipitate or worsen thiamine deficiency or WE in susceptible patients, so it is important that IV thiamine be given prior to or concurrent with IV fluids containing glucose.
Key:JZRWCGZRTZMZEH-UHFFFAOYSA-NThiamine, also known as thiamin or vitamin B 1, is a found in food, and manufactured as a. Food sources of thiamine include, and some meats and fish. Removes much of the thiamine content, so in many countries and are with thiamine.
Supplements and medications are available to treat and prevent and disorders that result from it, including. Other uses include the treatment of. They are typically taken, but may also be given by or.Thiamine supplements are generally well tolerated., including, may occur when repeated doses are given by injection. Thiamine is in the family. It is an, which cannot be made in the body. Thiamine is required for including that of, and.Thiamine was discovered in 1897, was the first vitamin to be isolated in 1926, and was first made in 1936. It is on the, the most effective and safe medicines needed in a.
Thiamine is available as a, and as an. The wholesale cost in the (as of 2016) is about US$2.17 per one gm vial. In the United States a month's supply of a multivitamin containing thiamine is less than US$25. See also:Thiamine is used to treat which when severe can prove fatal. In less severe cases, non-specific signs include, weight loss, irritability and confusion. Well-known disorders caused by thiamine deficiency include, African Seasonal Ataxia, and.In Western countries, thiamine deficiency is seen mainly in chronic alcoholism.
Thiamine deficiency is often present in. Also at risk are older adults, persons with HIV/AIDS or diabetes, and persons who have had. Varying degrees of thiamine deficiency have been associated with the long-term use of high doses of diuretics, particularly in the treatment of. Prenatal supplementation. See also:Women who are pregnant or lactating require more thiamine.
For pregnant and lactating women, the consequences of thiamine deficiency are the same as those of the general population but the risk is greater due to their temporarily increased need for this nutrient. In pregnancy, this is likely due to thiamine being preferentially sent to the fetus and placenta, especially during the third trimester.
For lactating women, thiamine is delivered in breast milk even if it results in thiamine deficiency in the mother. Pregnant women with are also at an increased risk for thiamine deficiency due to losses when vomiting.Thiamine is an important aspect for not only mitochondrial membrane development, but also synaptosomal membrane function. It has also been suggested that thiamine deficiency plays a role in the poor development of the infant brain that can lead to (SIDS). Other uses Thiamine is a treatment for some types of. Adverse effects Thiamine is generally well-tolerated and non-toxic when ingested. Some adverse side effects have been reported when thiamine is given including allergic reactions,. Chemistry Thiamine is a colorless with a 12 17 4.
Its structure consists of an and a ring linked by a. The thiazole is substituted with methyl and hydroxyethyl side chains. Thiamine is in, and and practically insoluble in less polar. It is stable at acidic pH, but is unstable in alkaline solutions. Thiamine, which is a, is used by enzymes to catalyze in vivo.
Thiamine is unstable to heat, but stable during frozen storage. It is unstable when exposed to ultraviolet light. Thiamine reacts strongly in. Biosynthesis. A 3D representation of the with thiamine boundComplex thiamine biosynthesis occurs in bacteria, some protozoans, plants, and fungi. The and moieties are biosynthesized separately and then combined to form (ThMP) by the action of thiamine-phosphate synthase.
The biosynthetic pathways may differ among organisms. In and other, ThMP may be phosphorylated to the (ThDP) by a (ThMP + ATP → ThDP + ADP, EC 2.7.4.16). In most bacteria and in, ThMP is hydrolyzed to thiamine, which may then be pyrophosphorylated to ThDP by (thiamine + ATP → ThDP + AMP, EC 2.7.6.2).The biosynthetic pathways are regulated. If there is sufficient thiamine present in the cell then the thiamine binds to the for the enzymes that are required in the pathway and prevents their. If there is no thiamine present then there is no inhibition, and the enzymes required for the biosynthesis are produced. The specific riboswitch, the (or ), is the only riboswitch identified in both eukaryotic and organisms. Nutrition Occurrence in foods Thiamine is found in a wide variety of processed and whole foods, with edible seeds, and, such as, having among the highest contents.The salt thiamine mononitrate, rather than thiamine hydrochloride, is used for food fortification, as the mononitrate is more stable, and does not absorb water from natural humidity (is non-hygroscopic), whereas thiamine hydrochloride is hygroscopic.
When thiamine mononitrate dissolves in water, it releases nitrate (about 19% of its weight) and is thereafter absorbed as the thiamine cation.United StatesAge groupRDA (mg/day)Tolerable upper intake levelInfants 0–6 months0.2.NDInfants 6–12 months0.3.1–3 years0.54–8 years0.69–13 years0.9Females 14–18 years1.0Males 14+ years1.2Females 19+ years1.1Pregnant/lactating females 14–501.4. Adequate intake for infants, as an RDA has yet to be establishedEuropean Food Safety AuthorityAge groupAdequate Intake (mg/MJ)Tolerable upper limitAll persons 7 months+0.1NDDietary recommendations In the U.S.
The Estimated Average Requirements (EARs) and Recommended Dietary Allowances (RDAs) for thiamine were updated in 1998, by the Institute of Medicine now known as the (NAM).The (EFSA) refers to the collective set of information as, with Population Reference Intake (PRI) instead of RDA, and Average Requirement instead of EAR. AI and UL defined the same as in United States. For women (including those pregnant or lactating), men and children the PRI is 0.1 mg thiamine per megajoule (MJ) of energy consumed. As the conversion is 1 MJ = 238.8 kcal, an adult consuming 2388 calories should be consuming 1.0 mg thiamine.
This is slightly lower than the U.S. The EFSA reviewed the same safety question and also reached the conclusion that there was not sufficient evidence to set a UL for thiamine.To aid with adequate micronutrient intake, pregnant women are often advised to take a daily. While micronutrient compositions vary among different vitamins, a typical prenatal vitamin contains around 1.5 mg of thiamine.For U.S.
Food and dietary supplement labeling purposes the amount in a serving is expressed as a percentage of Daily Value (%DV). For thiamine labeling purposes 100% of the Daily Value was 1.5 mg, but as of May 27, 2016 it was revised to 1.2 mg to bring it into agreement with the RDA. A table of the old and new adult Daily Values is provided at. The original deadline to be in compliance was July 28, 2018, but on September 29, 2017 the FDA released a proposed rule that extended the deadline to January 1, 2020 for large companies and January 1, 2021 for small companies. Antagonists Thiamine in foods can be degraded in a variety of ways., which are added to foods usually as a preservative, will attack thiamine at the methylene bridge in the structure, cleaving the pyrimidine ring from the thiazole ring.
The rate of this reaction is increased under acidic conditions. Thiamine is degraded by thermolabile (present in raw fish and shellfish ). Some thiaminases are produced by bacteria. Bacterial thiaminases are cell surface enzymes that must dissociate from the membrane before being activated; the dissociation can occur in ruminants under acidotic conditions. Rumen bacteria also reduce sulfate to sulfite, therefore high dietary intakes of sulfate can have thiamine-antagonistic activities.Plant thiamine antagonists are heat-stable and occur as both the ortho- and para-hydroxyphenols. Some examples of these antagonists are,. These compounds interact with the thiamine to oxidize the thiazole ring, thus rendering it unable to be absorbed.
Two flavonoids, and, have also been implicated as thiamine antagonists. Food fortification.
Main article:grain removes its bran and germ, and thus subtracts its naturally occurring vitamins and minerals. In the United States, B-vitamin deficiencies became common in the first half of the 20th century due to white flour consumption. The American Medical Association successfully lobbied for restoring these vitamins by enrichment of grain, which began in the US in 1939. The UK followed in 1940 and Denmark in 1953. As of 2016, about 85 countries had passed legislation mandating fortification of wheat flour with at least some nutrients, and 28% of industrially milled flour was fortified, often with thiamine and other B vitamins. Absorption and transport Absorption Thiamine is released by the action of and in the upper small intestine.
At low concentrations, the process is carrier-mediated. At higher concentrations, absorption also occurs via. Active transport is greatest in the jejunum and ileum, but it can be inhibited by alcohol consumption. Decline in thiamine absorption occurs at intakes above 5 mg/day. On the serosal side of the intestine, discharge of the vitamin by those cells is dependent on Na +-dependent ATPase. Bound to serum proteins The majority of thiamine in is bound to proteins, mainly. Approximately 90% of total thiamine in blood is in.
A specific binding protein called thiamine-binding protein (TBP) has been identified in rat serum and is believed to be a hormone-regulated carrier protein important for tissue distribution of thiamine. Cellular uptake Uptake of thiamine by cells of the blood and other tissues occurs via active transport and passive diffusion. About 80% of intracellular thiamine is phosphorylated and most is bound to proteins. Two members of the SLC gene family of transporter proteins, SLC19A2 and SLC19A3, are capable of the thiamine transport.
In some tissues, thiamine uptake and secretion appears to be mediated by a soluble thiamine transporter that is dependent on Na + and a transcellular proton gradient. Tissue distribution Human storage of thiamine is about 25 to 30 mg, with the greatest concentrations in skeletal muscle, heart, brain, liver, and kidneys. And free (unphosphorylated) thiamine is present in plasma, milk, and, it is presumed, all.
Unlike the highly phosphorylated forms of thiamine, ThMP and free thiamine are capable of crossing cell membranes. Calcium and Magnesium have been shown to affect the distribution of thiamine in the body and Magnesium deficiency has been shown to aggravate thiamine deficiency. Thiamine contents in human tissues are less than those of other species. Excretion Thiamine and its acid metabolites (2-methyl-4-amino-5-pyrimidine carboxylic acid, 4-methyl-thiazole-5-acetic acid, and thiamine acetic acid) are excreted principally in the urine.
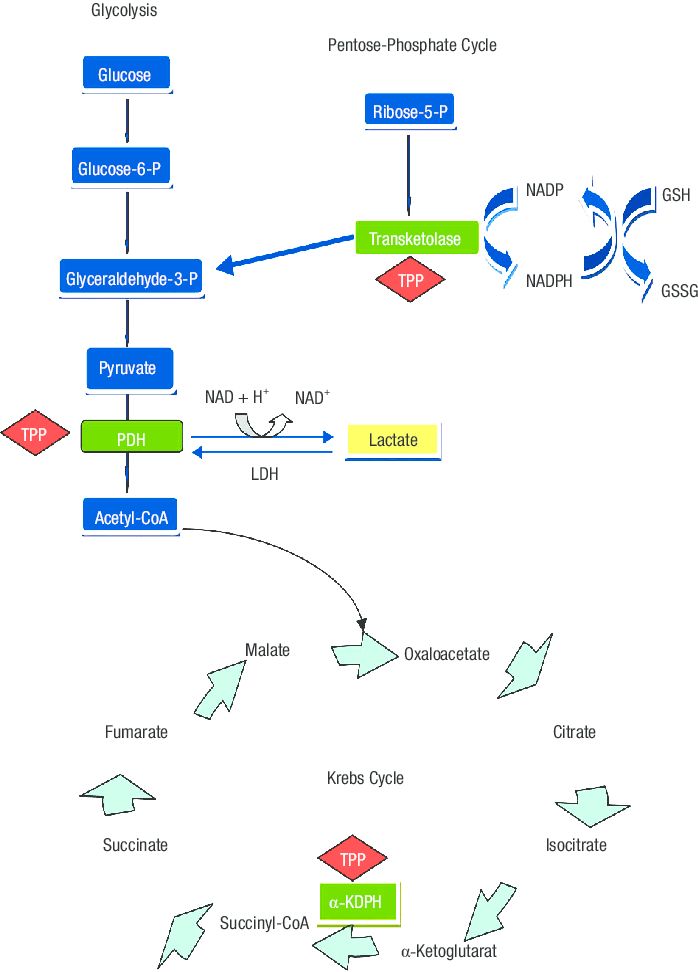
Function Its derivatives are involved in many cellular processes. The best-characterized form is (TPP), a in the of sugars and amino acids.
In yeast, TPP is also required in the first step of. All organisms use thiamine, but it is made only in bacteria, fungi, and plants. Animals must obtain it from their diet, and thus, for humans, it is an.
Insufficient intake in birds produces a characteristic.Thiamine is usually considered as the transport form of the vitamin. There are five known natural thiamine phosphate derivatives: (ThMP), (ThDP), also sometimes called (TPP), (ThTP), and the recently discovered (AThTP), and (AThDP). While the coenzyme role of thiamine diphosphate is well-known and extensively characterized, the non-coenzyme action of thiamine and derivatives may be realized through binding to a number of recently identified proteins which do not use the catalytic action of thiamine diphosphate Thiamine diphosphate No physiological role is known for thiamine monophosphate (ThMP); however, the diphosphate is physiologically relevant. The synthesis of (ThDP), also known as thiamine pyrophosphate (TPP) or cocarboxylase, is catalyzed by an enzyme called according to the reaction thiamine + ATP → ThDP + AMP (EC 2.7.6.2). ThDP is a for several enzymes that catalyze the transfer of two-carbon units and in particular the ( and subsequent conjugation with ) of 2-oxoacids (alpha-keto acids). Examples include:. Present in most species.
and 2- (also called ). Present in some species:. (in ). several additional enzymesThe enzymes, (PDH), and 2- (OGDH) are all important in. The cytosolic enzyme transketolase is a key player in the, a major route for the biosynthesis of the pentose. The mitochondrial PDH and OGDH are part of biochemical pathways that result in the generation of (ATP), which is a major form of energy for the cell.
PDH links glycolysis to the citric acid cycle, while the reaction catalyzed by OGDH is a rate-limiting step in the. In the nervous system, PDH is also involved in the production of acetylcholine, a neurotransmitter, and for myelin synthesis. Thiamine triphosphate (ThTP) was long considered a specific neuroactive form of thiamine, playing a role in chloride channels in the neurons of mammals and other animals, although this is not completely understood. However, recently it was shown that ThTP exists in, and suggesting a much more general cellular role.
In particular in, it seems to play a role in response to amino acid starvation. Adenosine thiamine triphosphate (AThTP) or thiaminylated adenosine triphosphate has recently been discovered in, where it accumulates as a result of carbon starvation. Coli, AThTP may account for up to 20% of total thiamine. It also exists in lesser amounts in, roots of higher plants and animal tissue. Adenosine thiamine diphosphate (AThDP) or thiaminylated adenosine diphosphate exists in small amounts in vertebrate liver, but its role remains unknown. History.
Some contributors to the discovery of thiamine. Thiamine was the first of the water-soluble vitamins to be described, leading to the discovery of more and to the notion of.In 1884, (1849–1920), a surgeon general in the, rejected the previous for beriberi and hypothesized that the disease was due to insufficiencies in the diet instead. Switching diets on a navy ship, he discovered that replacing a diet of white rice only with one also containing barley, meat, milk, bread, and vegetables, nearly eliminated beriberi on a nine-month sea voyage. However, Takaki had added many foods to the successful diet and he incorrectly attributed the benefit to increased nitrogen intake, as vitamins were unknown substances at the time. The Navy was not convinced of the need for so expensive a program of dietary improvement, and many men continued to die of beriberi, even during the of 1904–5. Not until 1905, after the anti-beriberi factor had been discovered in (removed by ) and in barley bran, was Takaki's experiment rewarded by making him a baron in the Japanese peerage system, after which he was affectionately called 'Barley Baron'.The specific connection to grain was made in 1897 by (1858–1930), a military doctor in the Dutch Indies, who discovered that fowl fed on a diet of cooked, polished rice developed paralysis, which could be reversed by discontinuing rice polishing.
He attributed beriberi to the high levels of starch in rice being toxic. He believed that the toxicity was countered in a compound present in the rice polishings. An associate, (1865–1944), correctly interpreted the connection between excessive consumption of polished rice and beriberi in 1901: He concluded that rice contains an essential nutrient in the outer layers of the grain that is removed by polishing. Eijkman was eventually awarded the in 1929, because his observations led to the discovery of vitamins.In 1910 a Japanese scientist first isolated the compound which he described as aberic acid. In translation from the Japanese paper in which it was claimed to be a new finding this claim was omitted.
In 1911 a Polish biochemist isolated the substance from rice bran (the modern thiamine) that he called a 'vitamine' (on account of its containing an amino group. However, Funk did not completely characterize its chemical structure. Dutch chemists, (1884–1962) and his closest collaborator (1889–1957), went on to isolate and crystallize the active agent in 1926, whose structure was determined by (1886–1965), a US chemist, in 1934. Thiamine was named by the Williams team as 'thio' or “sulfur-containing vitamin”, with the term 'vitamin' coming indirectly, by way of Funk, from the amine group of thiamine itself (by this time in 1936, vitamins were known to not always be amines, for example, vitamin C). Thiamine was synthesized in 1936 by the Williams group.Thiamine was first named 'aneurin' (for anti-neuritic vitamin). Sir, in Oxford, introduced thiamine-deprived pigeons as a model for understanding how thiamine deficiency can lead to the pathological-physiological symptoms of beriberi.
Indeed, feeding the pigeons upon polished rice leads to an easily recognizable behavior of head retraction, a condition called. If not treated, the animals died after a few days.
Administration of thiamine at the stage of opisthotonos led to a complete cure within 30 minutes. As no morphological modifications were observed in the brain of the pigeons before and after treatment with thiamine, Peters introduced the concept of a biochemical lesion.When Lohman and Schuster (1937) showed that the diphosphorylated thiamine derivative (thiamine diphosphate, ThDP) was a cofactor required for the oxydative decarboxylation of pyruvate, a reaction now known to be catalyzed by, the mechanism of action of thiamine in the cellular metabolism seemed to be elucidated. At present, this view seems to be oversimplified: pyruvate dehydrogenase is only one of several enzymes requiring thiamine diphosphate as a cofactor; moreover, other thiamine phosphate derivatives have been discovered since then, and they may also contribute to the symptoms observed during thiamine deficiency. Lastly, the mechanism by which the thiamine moiety of ThDP exerts its coenzyme function by proton substitution on position 2 of the was elucidated by in 1958.
See also.References.
